EXAMPLE: PROTEIN KINASES
The cAMP-dependent protein kinase as a model system for interaction studies
The cAMP-dependent protein kinase (PKA) is a heterotetramer consisting of two monomeric catalytic (C-) and a dimer of regulatory (R-) subunits. The activation of the inactive holo enzyme complex occurs upon binding of the second messenger cyclic AMP (cAMP) to the cAMP binding sites A and B in the regulatory subunits thus inducing a conformational change and a release of active catalytic subunits which are able to phosphorylate substrate proteins in the cell.
The heat stable protein kinase inhibitor PKI is a thermostable pseudo subtrate inhibitor and binds to the catalytic subunit of cAMP-dependent protein kinase PKA-C(alpha). The interaction between PKI and the catalytic subunit PKA-C(alpha) causes the export from the nucleus to the cytoplasm thus affecting transcriptional activity in the cell.
Figure: Interaction analysis of the heatstable protein kinase inhibitor PKI immobilised on a Biacore sensor surface with the catalytic subunit of cAMP-dependent protein kinase PKA-C(alpha) as analyte in solution. Assuming a 1:1 binding model the non linear data analysis after subtraction of the reference signal yielded the following rate constants [Zimmermann et al., J. Biol. Chem. (1999), 274, 5370-5378]:
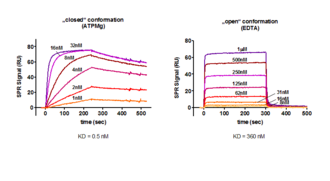
kass = 1.5 x106 M-1 s-1; kdiss = 7.6 x 10-4 s-1; KD = 0.5 nM
In the absence of ATP/Mg in a running buffer containing ETDA, PKA changes its conformation from a closed to an open state, resulting in a completely different binding kinetics for PKI. Here the binding affinity is lowered by a factor of 720 to KD = 360 nM.
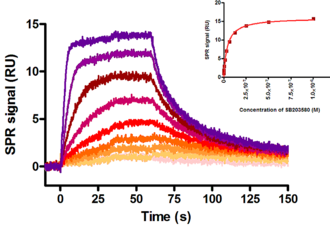
Figure: Interaction analysis of the MAP kinase p38 / SAPK2 alpha immobilised on a Biacore S51 sensor surface with the MAP kinase inhibitor SB203580 as analyte in solution. Assuming a 1:1 binding model the non linear data analysis after reference subtraction yielded the following rate constants:
kass = (1,9 ± 1,5) 106 M-1 s-1; kdiss = (5,1 ± 2,2) 10-2 s-1; KD = (32,1 ± 10,5) nM
For more information about our Biacore services especially in the area of protein kinase research please refer to one of our competent application specialists. We will be glad to prepare a suitable working plan and quotation for your specific requirements.
Tel.: +49 (0) 561-804 4661 | Fax: +49 (0) 561-804 4665 | info@biaffin.de
We value your privacy
In order to optimize our website for you and to be able to continuously improve it, we use cookies. Further information is available in our Data Protection Statement. Here you can find our Legal Information.