BIA technology for hit validation and lead optimisation
Are you interested in getting insights into how structural changes affect kinetic data of your optimized lead compound ?
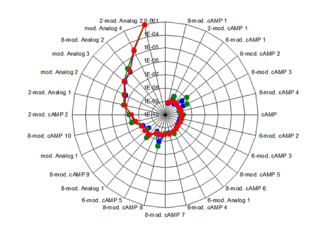
Biosensor sytems provide detailed information for QSAR (Quantitative Structure Activity Relationship) studies.
Hit validation is needed to determine whether a molecule identified in a screen or assay will eventually lead to a drug. Therefore secondary assays generating data about potency, selectivity and functional biochemical activity have to be performed. Lead optimisation is an iterative process involving computer-assisted molecular modelling, chemical synthesis of new compounds and functional assays.
BIA technology has an enormous capability for the rapid confirmation of hits from high throughput screens by a comprehensive kinetic characterisation of potential lead compounds. Informations about affinities, rates of association and dissociation in complex formation and binding stoichiometries are valuable for a ranking and optimisation of lead compounds. Even compounds binding with low affinity or transient kinetics, often found in early phases of drug development, can accurately be identified.
BIA technology provides detailed information how structural changes affect association and dissociation rates and allow a quantitative structural and functional activity relationship analysis (QSAR) in drug discovery processing.
Our service:
- binding level screen of potential lead compounds
- kinetic characterization of of lead compound-target interaction for structure activity relationships analysis
Please contact our application specialists for getting detailed information and an individual quote.
Tel.: +49 (0) 561-804 4661 | Fax: +49 (0) 561-804 4665 | info@biaffin.de
We value your privacy
In order to optimize our website for you and to be able to continuously improve it, we use cookies. Further information is available in our Data Protection Statement. Here you can find our Legal Information.